Mice are rodents, and their small size and rapid reproductive cycle makes them easy to maintain and breed in the laboratory. These factors have contributed to them becoming the most widely used laboratory animal, with over 80 million mice used in research worldwide. A large number of different strains of mouse are available. The mouse genome has been sequenced, and this species was one of the first mammals to undergo genetic manipulation to produce transgenic animals.
Basic biology and anatomy
Laboratory mice have been selectively bred from the wild house mouse, (Mus musculus) and retain many of the biological features of this wild rodent (Latham and Mason, 20041). They share common anatomical features with other terrestrial mammals, including humans, but have evolved some specific adaptations.
Mice, in common with all rodents, have continuously growing incisor teeth (at a rate of 0.4mm/day) and can gnaw hard materials easily. Laboratory mice are usually fed a hard, pelleted diet, composed of vegetable materials. The small size of mice requires greater energy expenditure to maintain body temperature and homeostasis than in larger species, so mice do not cope well with interruptions to their food supply, and food should always be freely available.
Like all rodents, mice have a simple, single-chambered stomach, divided into glandular and non-glandular sections. Mice cannot vomit. Their gastrointestinal tract is similar to other mammals but has a relatively large caecum, in which cellulose is digested. Since the caecum is located at the end of the small intestine, at the junction of the ileum and colon, little absorption of nutrients can occur. To circumvent this problem, mice ingest faecal pellets containing the material from the caecum (caecotrophy). This adaptation is shown by many laboratory rodents and also by rabbits, and can influence research protocols. For example, drug metabolites can be re-ingested and absorbed, and dietary components may be modified because of the addition of this further source of nutrients.
Special senses
Olfaction in mice is highly developed, and they use a wide array of chemical signals to gain information on the environment and on conspecifics. These may be both species-related molecules (pheromones) and mixtures of molecules specific to an individual animal (Mucignat-Caretta and Caretta, 20142). Mice excrete these signalling molecules in various body fluids (urine, tears, saliva, milk, and possibly other fluids) with urine being the most extensively studied. The emission of these chemical signals may modify the behaviour and/or the physiology of other animals, for example, aggression and attraction towards other mice.
Mice have a well-developed auditory system, with a hearing range of around 10-100kHz (human hearing range is approximately 20Hz to 20kHz) depending upon the intensity of the sound (Heffner and Heffner, 20073). Since mice can hear ultrasonic frequencies, it is important to identify sources of loud sounds in this frequency range in the animals’ environment. For example, computer monitors, air conditioning units and activities such as filling food hoppers can produce ultrasound at intensities of 60-90dB. It should also be appreciated that although mice vocalize at frequencies within the range audible to humans, they also make extensive ultrasonic calls. Some inbred strains show abnormalities and degenerative changes in their auditory systems. For example, C57BL/6 mice show deterioration of cochlear function that can be detected at around 6-7 weeks of age, and some albino strains have cochlear dysfunction from an early age (Henry and McGinn, 19924).
Mice are very sensitive to touch and have an extensive area of their sensory cortex devoted to processing information from their whiskers. They also have numerous guard hairs that are used to sense their environment.
The mouse visual system is well developed but visual acuity is poor in most strains of mice (Wong and Brown, 20065), as they rely primarily on olfaction, hearing, and their whiskers to sense their environment. However, vision is essential for some behaviours such as prey capture in daylight (Hoy et al, 20166). All albino strains have poor vision, and some pigmented strains have age-related blindness due, for example, to retinal degeneration (e.g. C3H strains and substrains are blind at the age of weaning, Chang et al, 20027). Mice perceive hue differently to humans, and their retinas are insensitive to red light but are sensitive to ultraviolet light.
Reproductive biology
Mice become sexually mature at the 6-7 week of age. Females have an oestrus cycle lasting 4-5 days. Mating usually results on the formation of a “vaginal plug” of material produced from the male accessory sex glands, and this can be used to confirm mating has taken place. Pregnancy lasts for 19-21 days, with the duration varying slightly between different strains. The fetuses can be detected using ultrasound or by gentle palpation of the abdomen by 7-10 days. As pregnancy progresses, the abdomen increases in size and the mammary glands, which extend from the inguinal region to the anterior thorax, increase in size.
The numbers of pups born varies considerably between strains and can range from only one or two to over twenty, but most mice have 10-16 pups in each litter (Fig. 2). Mouse pups are born hairless, blind and with their external ears closed, but develop rapidly (click to see images at 1 day old, 4 days old, 7 days old, 9 days old, 14 days old, 20 days old and 8 weeks) and are usually weaned at 3-4 weeks of age, depending upon their rate of growth and development.
Immediately after giving birth, mice mate again, so large numbers of offspring can be produced very rapidly.
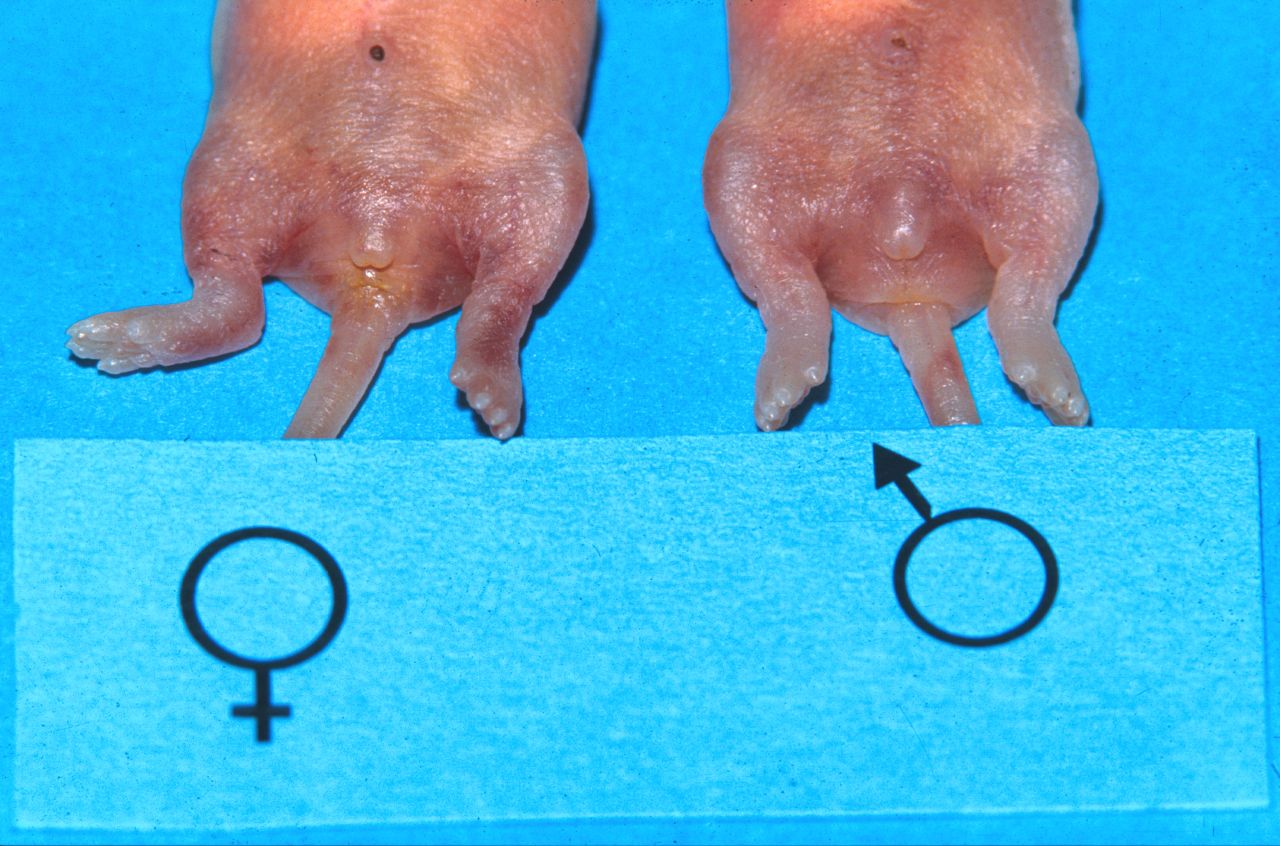
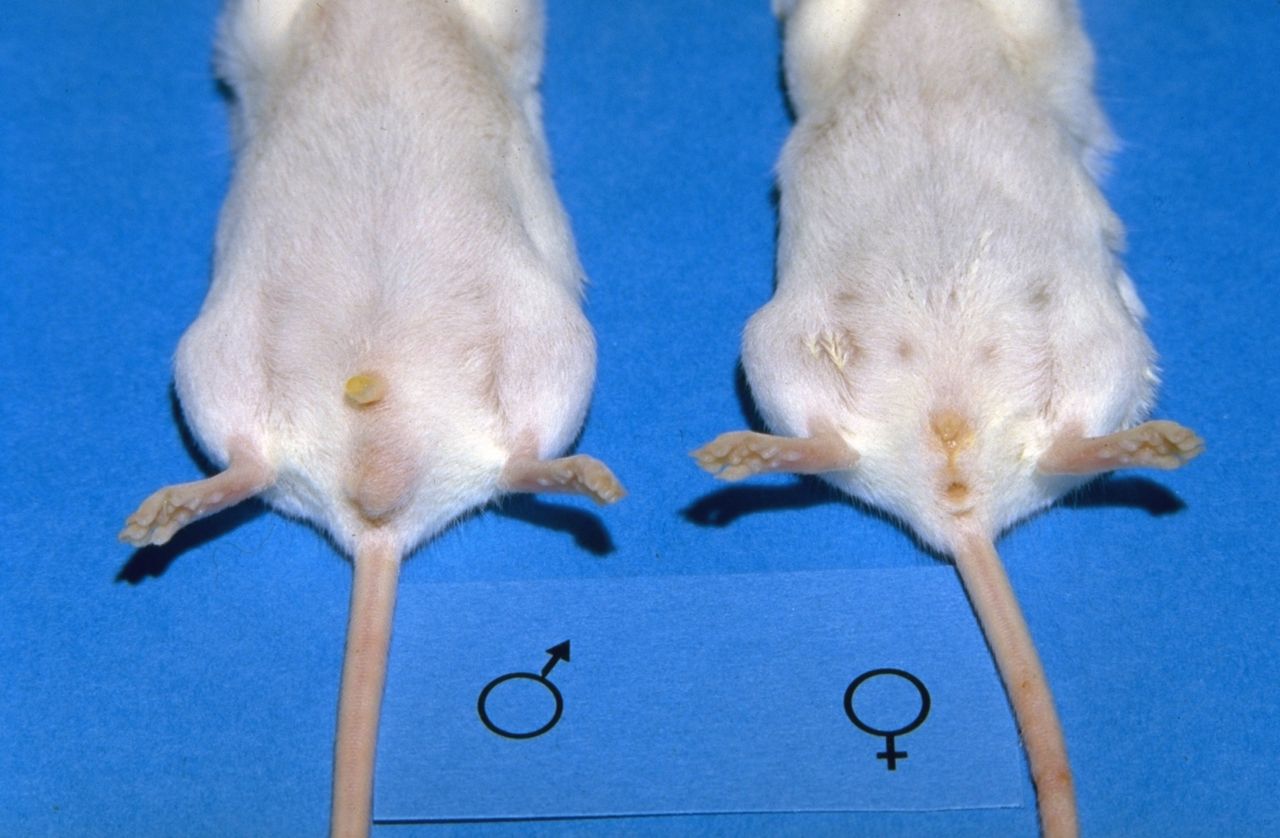
To avoid unwanted pregnancies, males and female offspring are housed separately after weaning. Determining the sex is relatively easy, as male mice have an obvious scrotum (Fig. 3). However they have an open inguinal canal, and the testes may be withdrawn back into the abdomen, reducing the size of the scrotum (Fig. 4). In younger mice, and in neonates, the sexes are differentiated by the distance between the external genital opening and the anus. This distance is larger in males than in females.
Mouse strains
A large number of different strains of mice have been developed for use in research, and many of these show variations in their physiology. Some have spontaneous genetic alterations which are of particular value for specific areas of research, for example, nude mice lack a thymus, and have no T cells, whereas SCID mice have no functional T or B cells. These mice require special husbandry and care to avoid exposing them to infectious agents since their immune systems are significantly compromised. Other strains require no specific husbandry conditions. The different strains of mice are usually categorized as either inbred or outbred. Inbred strains are established and maintained either by brother x sister or father x daughter matings and after approximately 20 generations become almost completely homozygous at all genetic loci. Inbred strains are named using an agreed international nomenclature. This is a combination of parent strain and substrain designations and may include an indication of the source of the strain. A parent strain is designated by a brief symbol made up of upper-case letters or numbers or combination of letters and numbers. For example, the most widely used strains of mice are C57BL, C3H, CBA, BALB/c, and AKR, and when given in full, these designations are extended to indicate the supplier – eg C57BL/6J (C57BL mice, substrain 6, from the Jackson Laboratory). Further information on mouse genetics is available online from the Jackson Laboratory .
In contrast to inbred strains, outbred strains, such as Swiss Webster and CD-1 mice, have more genetic variation within each strain. These strains are maintained by deliberate outbreeding to maintain this heterozygosity.
Transgenic and mutant mice
Large numbers of mice used in research have specific genetic alterations. These may have arisen spontaneously, either within an existing inbred strain (coisogenic strains) or by backcrossing a mouse with a particular mutation onto a specific inbred strain (congenic strains). Transgenic mice have foreign DNA intentionally inserted into their own genes, and this may result in alterations in gene expression – including over and under expression, or insertion of new genes. The technology to manipulate the mouse genome has become complex and sophisticated, and can now be used to manipulate multiple genetic characteristics rapidly (for example by use of CRISPR/Cas 9 techniques). Extensive information on these techniques can be found in the bibliography at the end of this section.
The phenotype of some GA lines is well established, and the effects on the animals’ welfare will have been identified. It may also be possible to alter housing, husbandry and research procedures to reduce the welfare consequences of the animals’ phenotype. In some circumstances, the effects of the genetic alteration will not have been established. Although it may be possible to predict the impact on the animals’ phenotype, unexpected effects can occur, so careful monitoring will be needed to detect these.
Behaviour
Mice are generally social animals but can show aggression, particularly towards unfamiliar animals. If housed together in single-sex groups (to prevent unwanted breeding) from shortly after weaning, stable social groups generally form, but these can break down and serious fight injuries can occur. Some strains are more likely to fight and cause injuries to cage mates.
Mice actively scent mark their environment, and olfaction plays a major role in communication, establishment of social hierarchies and in reproductive behaviour. Current standard methods of husbandry, which involve transferring animals to a cleaned cage at regular intervals, can disrupt this process and trigger aggression. Replacing nesting material back into cages can help reduce the stress associated with this process.
Mice are generally most active in the dark phase of their photoperiod, and also consume most food and water during this period. They actively explore their cage environment, climb, burrow and nest build if given appropriate substrates. They will also make use of housing structures placed in their cages (Fig. 5).
Providing a reasonably complex and enriched environment, which facilitates these normal behaviours, improves the welfare of caged mice and is easily achievable in a laboratory environment. Provision of nesting and bedding material (Fig. 6) is also important to enable mice to control their environmental temperatures. The preferred temperature of mice is well above the environmental temperatures specified by UK and EU codes of practice, but by building nests mice can increase the temperature locally to be closer to their thermoneutral zone.
Physiological values
| Mouse | |
|---|---|
| Adult body weight: male | 20-40g |
| Adult body weight: female | 25-40g |
| Birth weight | ¾-2g |
| Life span | 1½-3 years |
| Food consumption | 15g/100g/day |
| Water consumption | 15ml/100g/day |
| Breeding onset: male | 50 days |
| Breeding onset: female | 50-60 days |
| Cycle length | 4-5 days |
| Gestation period | 19-21 days |
| Postpartum estrus | Fertile |
| Litter size | 10-12 |
| Weaning age | 21-28 days |
| Breeding duration | 7-9 months |
| Commercial | 6-10 litters |
| Note that many of these values vary between different strains of mice, and also vary depending upon housing and husbandry conditions. | |
References
- Latham, N. and Mason, G., 2004. From house mouse to mouse house: the behavioural biology of free-living Mus musculus and its implications in the laboratory. Applied Animal Behaviour Science, 86(3), pp.261-289.
- Mucignat-Caretta, C. and Caretta, A., 2014. Message in a bottle: major urinary proteins and their multiple roles in mouse intraspecific chemical communication. Animal behaviour, 97, pp.255-263.
- Heffner, H.E. and Heffner, R.S., 2007. Hearing ranges of laboratory animals. Journal of the American Association for Laboratory Animal Science, 46(1), pp.20-22.
- Henry, K.R. and McGinn, M.D., 1992. The mouse as a model for human audition. Audiology, 31(4), pp.181-189.
- Wong, A.A. and Brown, R.E., 2006. Visual detection, pattern discrimination and visual acuity in 14 strains of mice. Genes, Brain and Behavior, 5(5), pp.389-403.
- Hoy, J.L., Yavorska, I., Wehr, M. and Niell, C.M., 2016. Vision drives accurate approach behavior during prey capture in laboratory mice. Current Biology, 26(22), pp.3046-3052.
- Chang, B., Hawes, N.L., Hurd, R.E., Davisson, M.T., Nusinowitz, S. and Heckenlively, J.R., 2002. Retinal degeneration mutants in the mouse. Vision research, 42(4), pp.517-525.