Handling and restraint
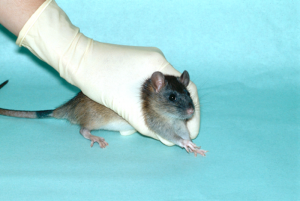
Rats are generally docile rodents provided they are approached and handled in a gentle manner. The rat should be picked up by grasping it around the shoulders (Fig. 1) and then lifted clear of its cage.
If the rat struggles when first restrained, it can usually be calmed by applying only minimal restraint and allowing the animal to rest on the handler’s sleeve. The animal can be allowed to move around with intermittent restraint being applied around the shoulders or to the base of the tail when it becomes necessary to adjust the position of the rat or to prevent escape.

After a short time, most rats will become more placid, and they may then be restrained more firmly for examination and to enable administration of substances or blood sampling to be carried out. If the rat appears aggressive, then it can be lifted by the base of the tail, transferred to a rough surface as described for the mouse and then grasped around the shoulders. It is rarely necessary to grasp a rat by the scruff, and this method of restraint is often resented by the animal unless it has become accustomed to the procedure.
The handler’s forefinger/thumb should be positioned under the mandible to prevent the animal from biting (Fig. 2). It is important that the grip around the rat’s shoulders should be sufficient to prevent its escape, but that only minimal pressure is applied to the thorax. Inexperienced handlers frequently tighten their grip if the rat struggles and this results in a severe impairment of chest movement. The rat will become severely distressed and struggle even more violently, will become cyanosed because they are unable to breathe adequately, and will almost invariably bite when released.
Oral administration
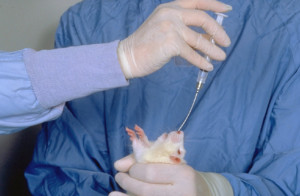
Whilst some substances are highly palatable and will be readily consumed when mixed with the drinking water or food, or even licked from the end of a syringe, unpalatable materials must be administered by stomach tube. Administration of accurate volumes of material will almost invariably require the use of a stomach tube. Suitable tubing can be made from 15 or 16 gauge needles, about 10-12 cm in length. The needle should have the short tip removed and a smooth ball of solder attached to its distal end. Alternatively, a rubber or polyethylene catheter, 3-4 f.g., may be used. A 16 gauge “Medicut” cannula (Sherwood Medical Industries Ltd., England) may also provide a suitable tube. Oral dosing needles are also available commercially. If using a plastic or rubber tube, a small gag may be necessary to prevent the rat from biting through the catheter.
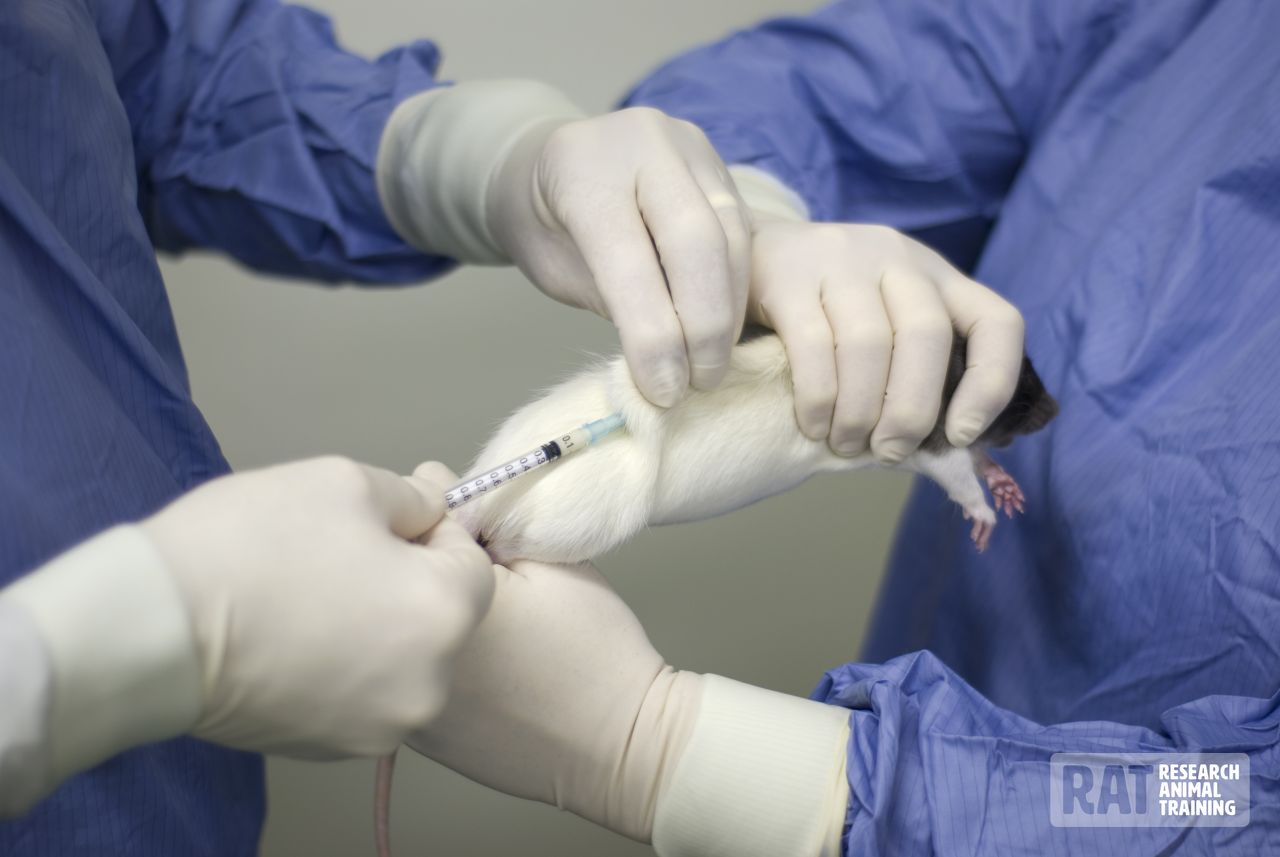
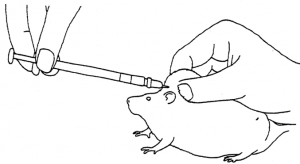
To pass the tube, the rat should be firmly restrained by grasping it by the skin of the back and neck; this ensures that the head and neck are extended and in line with the back (Figs. 3 and 4).
The tube can then be passed into the mouth via the interdental space and advanced gently, rotating the tube slightly to ease its passage into the oesophagus and/or into the stomach if necessary. In a conscious rat, it is unlikely that the tube will pass into the trachea.
Some rats will resent being restrained by the method described above and may struggle violently during the procedure. In these cases, it may be found simpler and more humane to anaesthetise the animal prior to passing the tube, especially if the procedure is being undertaken by one person. Following induction of anaesthesia, the tube can be passed easily, but when the rat is deeply anaesthetised it will be found that the tube occasionally enters the trachea. By keeping the animal at a light plane of anaesthesia the cough reflex is maintained and hence an indication will be given if the trachea is inadvertently intubated.
As an alternative to gavage, some materials may be consumed voluntarily in palatable mixtures (e.g. flavoured syrups, Leach et al, 20101, Corbett et al, 20122). Rats can also be trained to drink voluntarily from a syringeand this approach has been shown to be both effective and to result in accurate dosing (Atcha et al, 20103). Material can also be dosed using a small flexible catheter introduced only into the animal’s mouth. When possible, these approaches are to be preferred to dosing using a stomach tube, since they are not associated with complications, such as tracheal dosing or oesophageal rupture.
Injectable administration
Subcutaneous injection
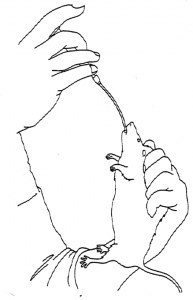
To carry out a subcutaneous injection, the rat should be restrained by grasping by the skin of the scruff, and the injection made into the area of skin “tented” by this procedure (Fig 5).
The needle should be positioned almost parallel to the skin and introduced through the skin and then on subcutaneously for most of its length.
If the rat has become accustomed to handling, then it is often possible to carry out subcutaneous injection into the skin overlying the neck with minimal restraint.
Intraperitoneal injection
For intraperitoneal injection, the rat is held by an assistant, with one leg held by the operator. The needle is advanced parallel to the line of the leg, and pushed through the abdominal wall into the peritoneal cavity; a loss of resistance to needle passage will be noted as the needle passes through the abdomen. By advancing the needle along the line of the leg, injection is made in an area that avoids the urinary bladder in the posterior abdomen, and the liver anteriorly. Although it is possible to inject into the gastrointestinal tract, this is a rare complication of intraperitoneal injection. It should be noted that most rats will urinate when restrained, and if this coincides with the injection of material intraperitoneally it can alarm an inexperienced operator!
Intramuscular injection
Suitable sites for intramuscular injection include the quadriceps, the posterior thigh and the triceps. Injection into the posterior thigh will cause the least discomfort to the animal, but injection into the fascial planes between the muscle bellies may be a problem when using this site. Care must be taken to inject into the most posterior part of the muscle mass, to avoid damage to the sciatic nerve. Injection into the quadriceps (Fig 6) often appears to cause more pain to the animal, although the pain can be minimised by slow injection of relatively small volumes of material. This latter route has the advantage that no major blood vessels or nerves are present in the main muscle bellies.
Intravenous injection
The most frequently used vessels for intravenous injection are the lateral tail veins, which can be readily seen in young rats. In older animals thickening of the skin over the tail makes it difficult to detect the vessel. Warming the tail by placing the rat under a heating lamp will dilate the vessel and facilitate venepuncture but great care must be taken to avoid excessive heat being applied. To inject into a tail vein the rat should be held by an assistant and the tail held by the operator. The vein can then be located and the needle advanced at a shallow angle. A loss of resistance will be felt as the needle enters the vein. To be certain that the needle is lying in the vein, a small quantity of material must be injected – if the needle tip is not lying within the vein a small bleb will be produced, and the needle must be repositioned. It is often easier to place the rat in a suitable restraining device since any sudden movements by the animal make injection difficult. Injections into other veins can only be undertaken after first inducing general anaesthesia. Full details of the use of the sublingual, femoral, jugular and dorsal metatarsal veins can be found in Waynforth and Flecknell, 19924.
Tutorial videos on the administration of substances in laboratory rats can be found at www.procedureswithcare.org.uk
References
- Leach, M.C., Forrester, A.R. and Flecknell, P.A., 2010. Influence of preferred foodstuffs on the antinociceptive effects of orally administered buprenorphine in laboratory rats. Laboratory animals, 44(1), pp.54-58.
- Corbett A, McGowin A, Sieber S, Flannery T, Sibbitt B.
A method for reliable voluntary oral administration of a fixed dosage (mg/kg) of chronic daily medication to rats. Laboratory animals. 2012 Oct;46(4):318-24.
- Atcha, Z., Rourke, C., Neo, A.H., Goh, C.W., Lim, J.S., Aw, C.C., Browne, E.R. and Pemberton, D.J., 2010. Alternative method of oral dosing for rats. Journal of the American Association for Laboratory Animal Science, 49(3), pp.335-343.
- Waynforth HB, Flecknell PA (1992) Experimental and Surgical Technique in the Rat, 2nd ed. London: Academic Press